Aquilo is an experienced CRO in 2D and 3D in vitro cell research.
A few of our in vitro testing assays and readouts are listed below.
However, additional read-outs depending on your needs, such as in vivo testing for COPD, asthma and lung fibrosis, can always be discussed.
Please contact us to find out our experience with your read-out of interest.
2D in vitro cell culture
Anti-inflammatory efficacy
Inhibition of cytokine (IL-1β, IL-13, TNF-α, others) or cigarette smoke induced responses, including:
- ELISA or Luminex analysis of pro-inflammatory cytokine release.
- Mucus seceretion (airway epithelial cells).
- Gene and protein expression for inflammatory markers such as COX-2.
- Activation of transcriptional mechanisms (e.g. STAT).
Anti-remodeling efficacy
Inhibition of growth factor (TGF-β, PDGF, WNT, others) induced responses, including:
- Cell proliferation.
- Cell migration or adhesion (xCELLigence).
- Myofibroblast and smooth muscle differentiation (lung fibroblasts, airway smooth muscle).
- Epithelial mesenchymal transition (EMT; epithelial cells).
- Angiogenesis.
- Gene and protein expression for matrix proteins, proliferation markers.
- Activation of transcriptional mechanisms (e.g. Smad, β-catenin, Sp1, others)
In addition, we can offer several possibilities for target validation in human samples.
3D in vitro cell culture
3D cell culture
- Air-liquid interface culture of airway epithelial cells
- Co-culture with fibroblasts or other cell types on request
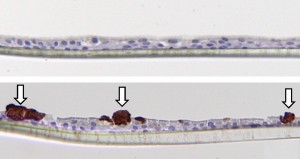
By using air liquid interfaces adequate study of epithelial cell function can be studied. with the use of this system your investigational compounds can be inserted into epithelial cells and the differentiation, goblet cell metaplasia and epithelial barrier function can be visualized.
Read-outs
- (Immune)histochemistry
- Gene expression
- ELISA of secreted factors in the medium.
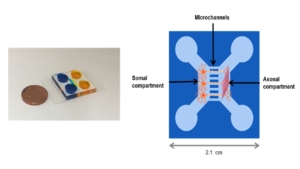
Lung on chip models
- Airway cholinergic neurons
- Airway smooth muscle cells
- Other cell types on request
This model is suitable for studies on neuro-effector interactions such as neuro-immune interactions and neuron-smooth muscle interactions and for studies into neuroplasticity in asthma.
Read-outs
- Neuronal density
- Calcium imaging
- Gene expression in neurons or ASM cells
This model was developed in collaboration with the University of Groningen and funded by a grant (114021505) from the ZonMW/Proefdiervrij Meer Kennis met Minder Dieren (Animal free – More Knowledge with Fewer Animals) program with co-financing from Boehringer Ingelheim, Lung Foundation Netherlands and Aquilo. See publication